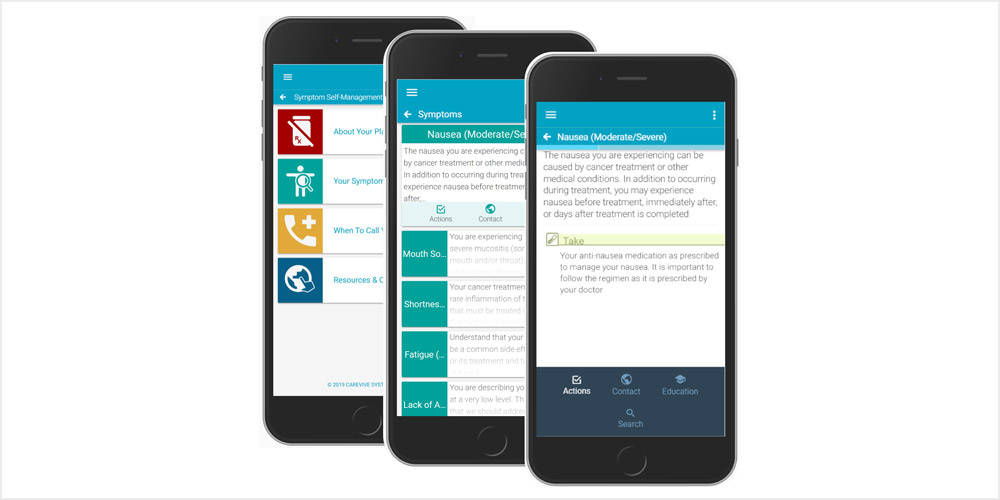
Carevive Systems had the opportunity to participate in the PRO-TECT Trial conducted by Ethan Basch and other medical leaders within the scientific research community. Carevive provided the study with the use of their 24/7 telephone triage protocols and self-management care plans for nursing triage support. A goal of the trial was to gain an understanding of the perspectives of care teams and patients regarding the routine collection of patient-reported outcomes (PROs) at cancer centers for symptom management purposes. Surveys that included the National Cancer Institute Patient-Reported Outcomes version of the Common Terminology Criteria for Adverse Events (PRO-CTCAE™) ePRO measure were administered on a weekly basis using automated phone/web systems, with alerts sent to nurses for management of severe or exacerbated symptoms. User feedback on the process was then elicited via surveys administered to clinicians and patients who participated in the trial.
Twenty-six U.S. community oncology practices participated with a total of 497 patients involved. Patients were specifically adults with advanced and metastatic cancers receiving treatment at the practices. In the study, feedback surveys were administered to patients after 3 months of participation completing PRO surveys. By the cutoff date of May 4, 2020, 497 patients participated in the PRO-TECT study and completed at least 3 months of participation.
Overall, patients reported finding the web-based self-reporting systems easy to understand and use between visits and felt that they enhanced the quality of care and communication with their care teams. In addition to improved discussions with care teams, patients reported feeling more in control through PRO reporting and would recommend to other patients. Clinicians who participated, including oncologists and nurses, also provided positive feedback, indicating excitement about being able to collect PROs in clinic. In total, 57 nurses and 39 oncologists completed feedback surveys after at least 6 months of using the system for patient care. Participating clinicians reported improved efficiency, improved discussions with patients, and improved overall quality of care delivered to patients. Additionally, providers indicated they’d like to use with future patients and would recommend to others.

Of note are benefits for both the care team and patients. Although there are concerns of the added workload from symptom alerts, nurses and providers do believe that the added insights are helpful in providing higher quality care. There is also a belief that there are efficiencies gained from less after-hour calls and fewer clinic interruptions that are typically caused when patients are hospitalized. Patients reported feeling more in control of their care and felt educational materials provided for severe or worsening symptoms were useful. The Carevive platform drives towards ensuring care teams and patients receive quality of life benefits throughout the patient care journey. Nurses are alerted for only those symptoms which require clinical intervention, allowing the care team to not be over-burdened by symptom alerts, and our goal is that through the use of machine learning, we will teach the platform on how to improve alerting functionality. Symptoms are caught earlier and patients receive education for any symptom reported reinforcing education on self-management strategies.
To read the full article, click here: https://ascopubs.org/doi/abs/10.1200/CCI.20.00081

